Home /
Expert Answers /
Chemistry /
consider-the-three-electronic-transitions-in-a-hydrogen-atom-shown-here-labeled-a-b-and-c-1-pa374
(Solved): Consider the three electronic transitions in a hydrogen atom shown here, labeled A, B, and C. (1) ( ...
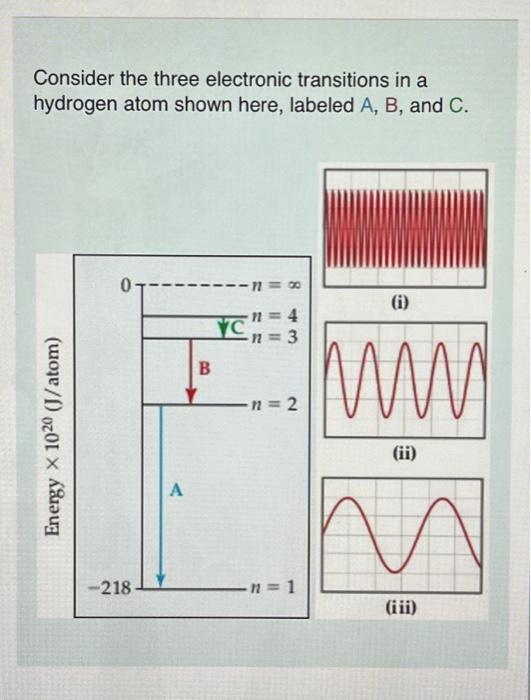
Consider the three electronic transitions in a hydrogen atom shown here, labeled A, B, and C. (1) (ii)
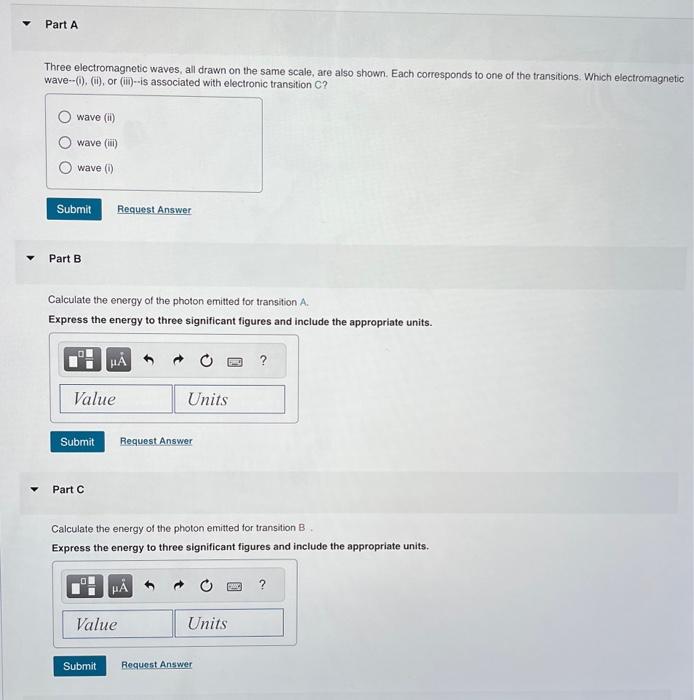
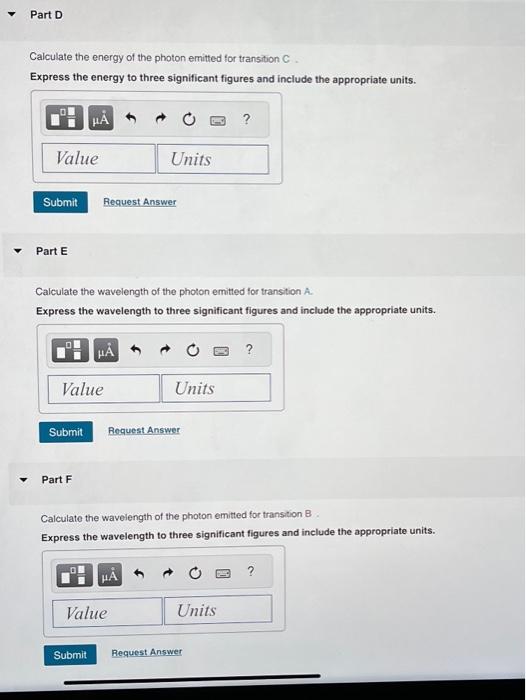
Three electromagnetic waves, all drawn on the same scale, are also shown. Each corresponds to one of the transitions. Which electromagnetic wave-(0), (ii), or (iii)--is associated with electronic transition C? Part B Calculate the energy of the photon emitted for transition . Express the energy to three significant figures and include the appropriate units. Part C Calculate the energy of the photon emitted for transition B Express the energy to three significant figures and include the appropriate units.
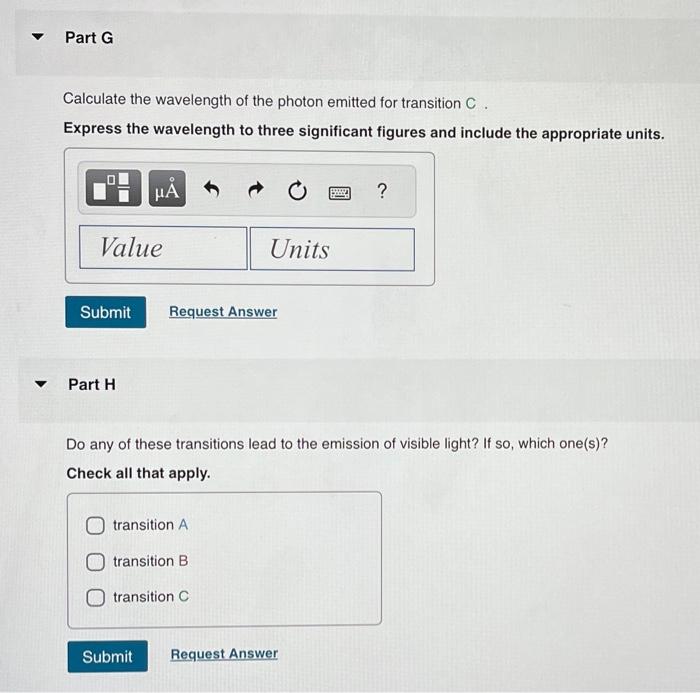
Calculate the energy of the photon emitted for transition C. Express the energy to three significant figures and include the appropriate units. Part E Calculate the wavelength of the photon emitted for transtion A. Express the wavelength to three significant figures and include the appropriate units. Part F Calculate the wavelength of the photon emitted for transition Express the wavelength to three significant figures and include the appropriate units.
Calculate the wavelength of the photon emitted for transition C. Express the wavelength to three significant figures and include the appropriate units. Part H Do any of these transitions lead to the emission of visible light? If so, which one(s)? Check all that apply. transition B transition C