Home /
Expert Answers /
Chemistry /
a-chemical-reaction-takes-place-inside-a-flask-submerged-in-a-water-bath-the-water-bath-contains-3-pa951
(Solved): A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 3 ...

A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains of water at . During the reaction of heat flows out of the bath and into the flask. Calculate the new temperature of the water bath. You can assume the specific heat capacity of water under these conditions is - . Round your answer to 3 significant digits.
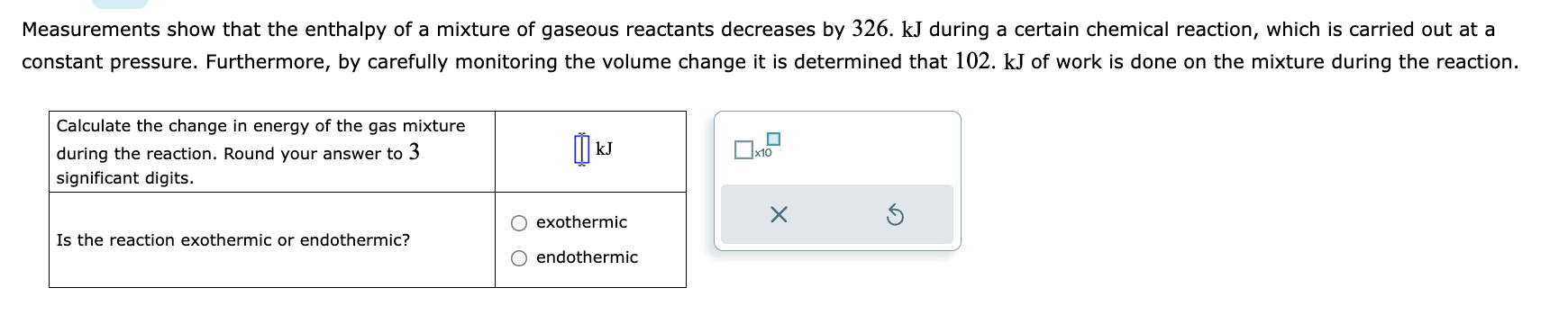
Measurements show that the enthalpy of a mixture of gaseous reactants decreases by 326 . during a certain chemical reaction, which is carried out at a constant pressure. Furthermore, by carefully monitoring the volume change it is determined that 102 . of work is done on the mixture during the reaction.
Expert Answer
We know, and, where Heat Mass of the substance Specific heat of the substance Change in temperature Final temperature Initial temperature