Home /
Expert Answers /
Chemistry /
for-many-purposes-we-can-treat-nitrogen-n-2-as-an-ideal-gas-at-temperatures-above-its-boiling-po-pa249
(Solved): For many purposes we can treat nitrogen (N_(2)) as an ideal gas at temperatures above its boiling po ...
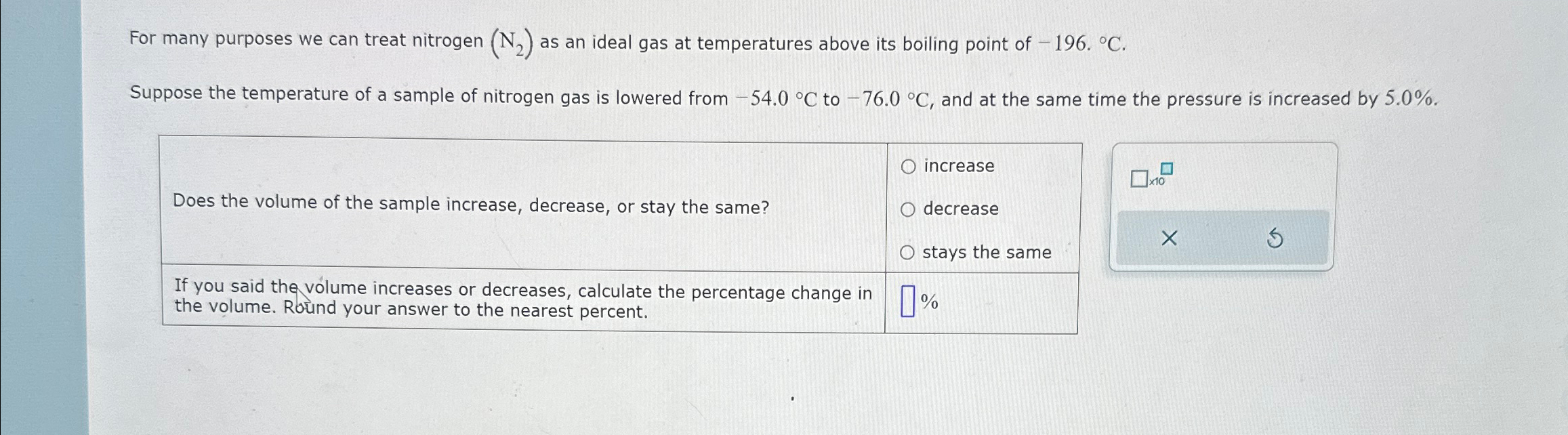
For many purposes we can treat nitrogen
(N_(2))as an ideal gas at temperatures above its boiling point of
-196.\deg C. Suppose the temperature of a sample of nitrogen gas is lowered from
-54.0\deg Cto
-76.0\deg C, and at the same time the pressure is increased by
5.0%. \table[[Does the volume of the sample increase, decrease, or stay the same?,increase],[,decrease],[If you said the volume increases or decreases, calculate the percentage change in,],[the volume. Round your answer to the nearest percent.,
%