Home /
Expert Answers /
Chemistry /
question-3-start-on-a-new-page-compounds-given-below-3-define-the-term-boiling-point-whic-pa259
(Solved): QUESTION 3 (Start on a new page.) compounds given below. 3. Define the term boiling point. Whic ...
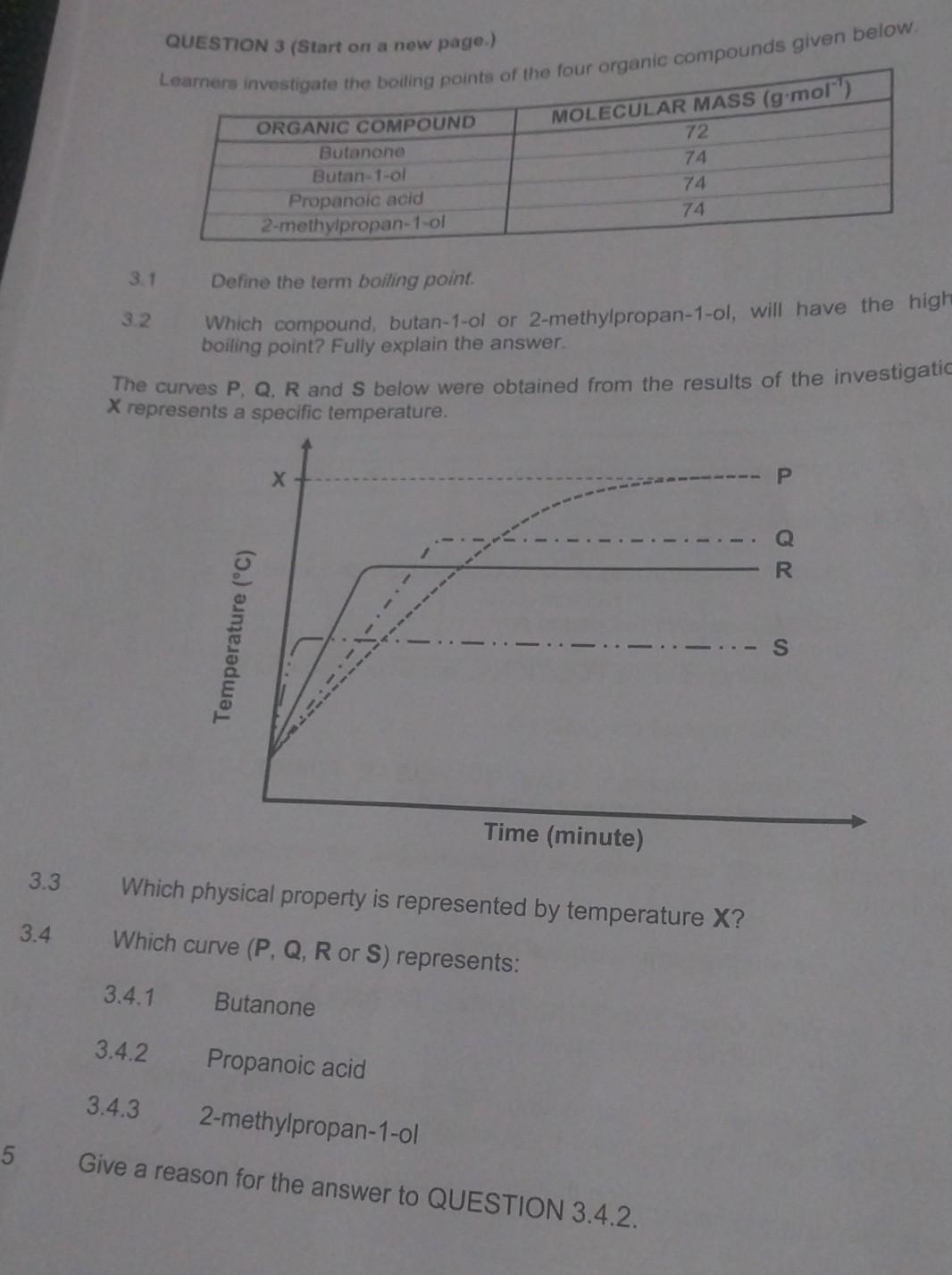
QUESTION 3 (Start on a new page.) compounds given below. 3. Define the term boiling point. Which compound, butan-1-ol or 2-methylpropan-1-ol, will have the high boiling point? Fully explain the answer. The curves and below were obtained from the results of the investigatio represents a specific temperature. W.3 Which physical property is represented by temperature ? 3.4 Which curve or represents: 3.4.1 Butanone 3.4.2 Propanoic acid -methylpropan-1-ol 5 Give a reason for the answer to QUESTION 3.4.2.
Expert Answer
3.1)The boiling point refers to the temperature at which a substance changes its phase from a liquid to a gas at a given pressure. It is the temperature at which the vapor pressure of the liquid equals the atmospheric pressure or the pressure applied to the system. At the boiling point, bubbles of vapor form within the liquid and rise to the surface, causing the liquid to vaporize. The boiling point is considered a characteristic property of a substance and can be influenced by factors such as atmospheric pressure and the presence of impurities.