Home /
Expert Answers /
Chemistry /
use-the-data-provided-to-calculate-the-delta-h-deg-for-the-following-reaction-2nh-3-g-3o-2-pa388
(Solved): Use the data provided to calculate the \Delta H\deg for the following reaction: 2NH_(3)(g)+3O_(2)( ...
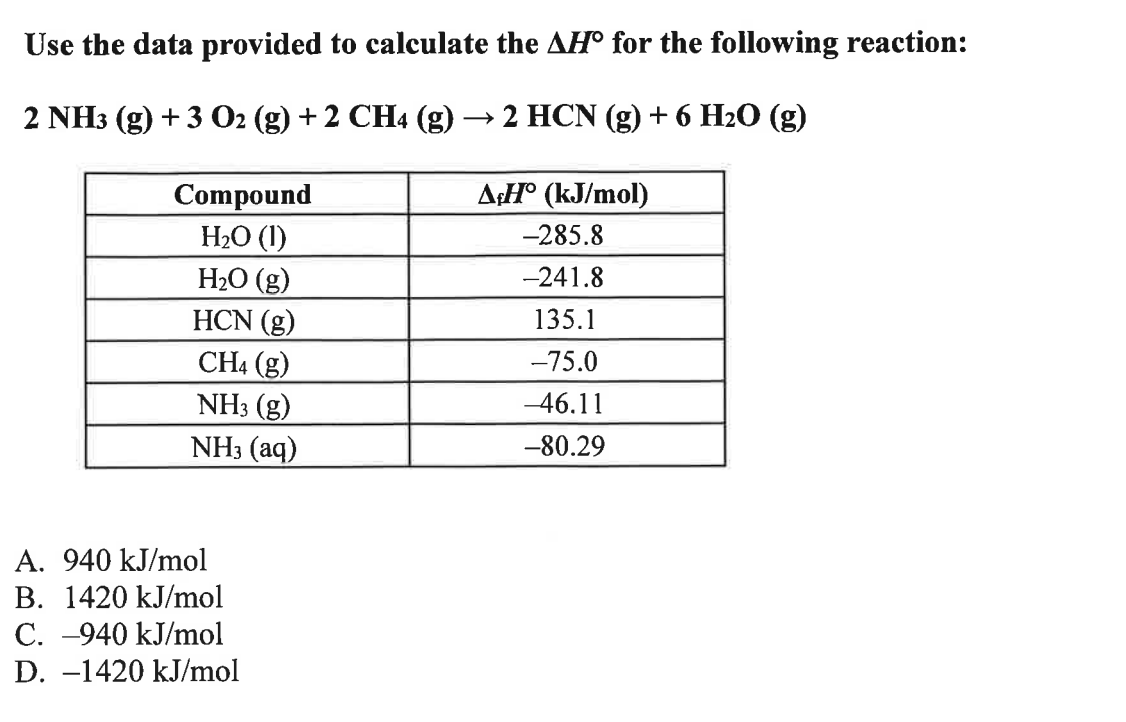
Use the data provided to calculate the
\Delta H\deg for the following reaction:
2NH_(3)(g)+3O_(2)(g)+2CH_(4)(g)->2HCN(g)+6H_(2)O(g)A.
940k(J)/(m)olB.
1420k(J)/(m)olC.
-940k(J)/(m)olD.
-1420k(J)/(m)ol