Home /
Expert Answers /
Chemistry /
use-the-references-to-access-important-values-if-needed-for-this-question-to-answer-the-questions-pa430
(Solved): Use the References to access important values if needed for this question. To answer the questions ...
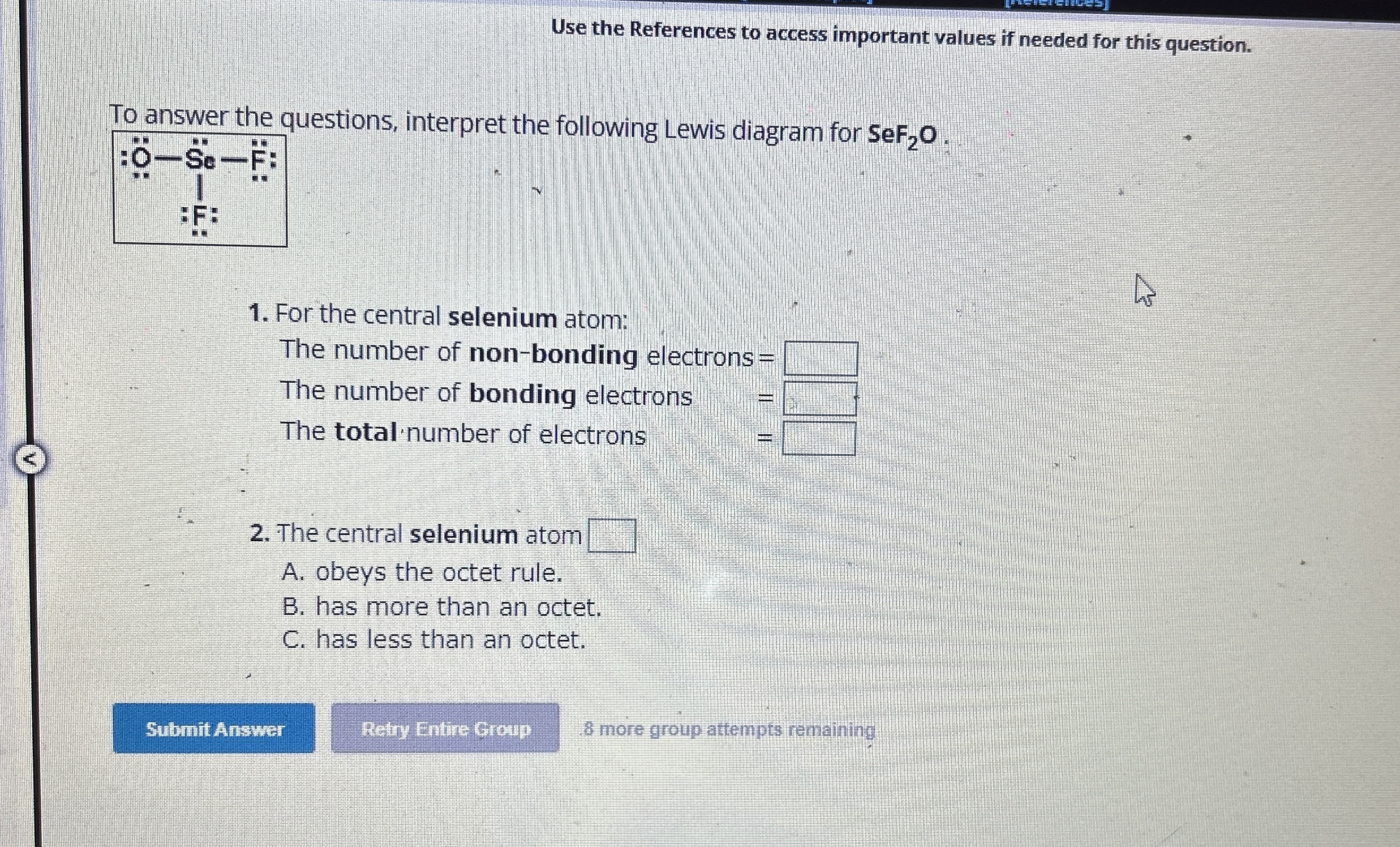
Use the References to access important values if needed for this question. To answer the questions, interpret the following Lewis diagram for
SeF_(2)O. For the central selenium atom: The number of non-bonding electrons
=
◻The number of bonding electrons
=
◻The total number of electrons
=
◻The central selenium atom
◻A. obeys the octet rule. B. has more than an octet. C. has less than an octet.
◻8 more group attempts remaining